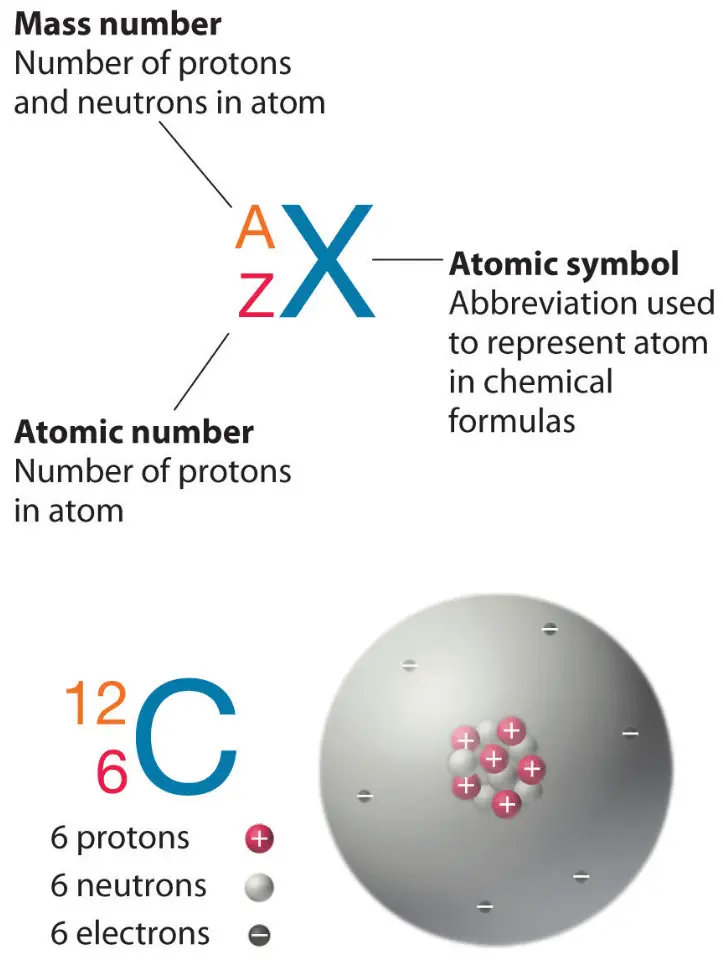
They are so small that for practical purposes they do not contribute to the mass of the atom. Electrons have a negative charge but are extremely small and have a mass only 1/1850 that of a proton or neutron. The third type of stable particle is the electron. Neutrons and protons constitute almost all of an atom’s mass. 
Although the mass of the neutron is slightly greater than that of a proton we can assign both of them the relative mass of 1 (1 atomic mass unit or amu). We refer to these atoms as being radioactive. Too many or too few neutrons may result in an atomic nucleus that is unstable and may decay to form other elements. Neutrons bind with protons in a way that helps stabilize the nucleus. Neutrons have no charge, have mass, and are also located in the nucleus of the atom. Protons are positively charged particles, have mass, and are located in the center, or nucleus of the atom. These are the protons, neutrons, and electrons. Physicists have succeeded in blasting atoms apart into dozens of different sub-atomic particles, however, only 3 of them are stable. Therefore all of the atoms that make up the element carbon have the same chemical properties. Chemical properties include the physical state of the element (gas, liquid, or solid), the types of bonds the element can form, how it reacts with other elements, etc. We can define an atom as the simplest particle of an element that has the chemical properties of that element. Inorganic Chemistry (Atomic Structure) AtomsĪll elements are composed of extremely small particles of matter called atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
